Technology to overcome therapeutic inertia
Clinical inertia or therapeutic inertia is a major health problem in type 2 diabetes (T2D). So much so that the American Diabetes Association (ADA) recently kicked off a multi-year campaign to identify the issues, address barriers and develop solutions (1). Therapeutic inertia is defined as “the failure to initiate or intensify therapy in a timely manner according to evidence-based clinical guidelines.” (2). The term therapeutic implies that both patients and healthcare providers (HCPs) are together part of the problem and the solution. A recent systematic review documented that on average it took more than 1 year from the start of treatment intensification until an A1C target was achieved (2). And in some studies, the range was more than 7 years! In the United States we are not meeting goals for diabetes management despite the multitude of new medications on the market to manage T2D and up-to-date clinical guidelines (3).
In a recent study, Clinical Inertia in a Randomized Trial of Telemedicine-Based Chronic Disease Management: Lessons Learned (4) Barton et al. describe a telemedicine intervention that was explicitly designed to address clinical inertia, yet did not succeed in meeting their goals. This study enrolled 359 African Americans with T2D, and randomized 182 to The Cholesterol, Hypertension, and Glucose Education (CHANGE) (5) study intervention. The CHANGE program used skilled nurses to (I) educate about self-management to promote healthy behaviors including medication taking and (II) supported treatment intensification by communicating with HCPs. Nurses engaged in monthly phone calls with patients and focused on 3 areas including educating about self-management, addressing psychosocial concerns and supporting behavior change. Every 3 months nurses electronically messaged HCPs to inform them about patient progress and suggested medication changes that may be indicated. They also offered to facilitate medication changes by communicating directly with patients.
Despite these resources and focused attempts to address inertia, at least 67% of encounters resulted in inappropriate, non-intensification of treatment even when A1C, blood pressure or cholesterol parameters were not at goal. In fact, A1C was typically not intensified unless the mean value was 9.7%. Also, during the study there were 31 participants (17%) who chose to decline intensification, despite not being at goal (4).
Why is this so?
There is an abundance of thought on why therapeutic inertia exists and yet studies have shown that HCPs and people living with diabetes (PWD) have different perceptions of the value and benefits of taking more medication. Some PWD view more medication as a sign that their condition is getting worse or they are failing (6). PWD may be reluctant to change medications feeling that they can and “should” be able to manage with behavior changes. The language we use in healthcare often blames and stigmatizes people with diabetes; often HCPs “threaten” patients with insulin if they don’t succeed as opposed to identifying insulin as a helpful tool in managing diabetes (7). Although forgetfulness, access to medication and costs are often cited by HCPs as being the main drivers of PWD not taking medications, in reality there is often a lack of trust between patients and providers, a lack of communication and a perceived lack of empathy from HCPs (8,9). Thus, HCPs make assumptions that their patient is not going to want to take the medication and often choose not to make the recommendation.
While this study attempted to minimize barriers to treatment intensification with the support of technology, there are opportunities now to learn from their experience. First, the original study began in 2008, over 10 years ago when telehealth solutions were just beginning and primarily used a telephonic-based system with home-based blood pressure monitors. Increasingly, with the introduction of smartphone technology, digital health tools with embedded analytic algorithms are providing more actionable real-time information for individuals, supporting focused conversations with patients and clinical decision support for HCPs. Digital therapeutics are demonstrating outcomes in chronic disease management for diabetes, mental health, and asthma and provide a potential for impacting therapeutic inertia (10).
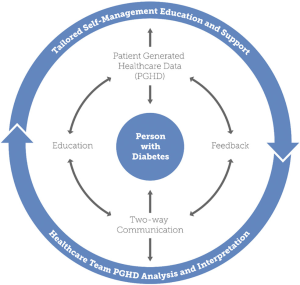
Newer research shows that improving outcomes is possible in clinical trials. In 2017, a systematic review of 25 review articles published since 2013 found that the utilization of technology in diabetes self-management education and support services resulted in significant reduction in A1C (11). Four key elements emerged as essential to improve outcomes: (I) communication, (II) patient-generated health data (PGHD), (III) education, and (IV) feedback. These elements are incorporated into the technology enabled self-management (TES) feedback loop (see Figure 1) (11).
How can future studies be designed to improve the outcomes?
In a similar telehealth remote patient monitoring (RPM) intervention designed to overcome clinical inertia, there was a small, but statistically significant improvement in A1C compared to control (12). Compared to the CHANGE study, this trial incorporated real time RPM combined with a structured self-monitoring of blood glucose (SMBG) approach called “checking in pairs” which empowered participants to perform daily experiments and learn how their blood glucose responded to changes in eating and activity (12). The CHANGE study did not report if and how remote monitoring data were used in the determination of treatment recommendations. In the RPM study, because nurses had real-time access to SMBG data, they were able to reach out to participants frequently and begin to discuss medication changes directly with patients in response to their daily experiment data. Nurses used a shared decision-making approach to explore medication choices thus building trust over time. In the RPM study, 30% of participants did change medications (12).
In addition, a systematic review showed that randomized clinical trials (RCTs) with telehealth RPM interventions that incorporated structured SMBG principles resulted in greater A1C improvement (13). Access to actionable, real-time PGHD, creates an opportunity for feedback and ultimately changes in therapy.
The TES feedback loop may provide a more granular way to design studies to include technology interventions (11). This framework can also be used to describe the technology components of a clinical intervention: incorporating 2-way communication, the use of and analysis of PGHD to tailor education and customize feedback.
How do we translate clinical trial outcomes into practice?
The paper by Barton et al. (4) focused on the key elements of the Chronic Care Model (14) (CCM) that include the engaged patient (“a”) and the proactive care team (“b”) that are essential in the achievement of outcomes (“c”). The a + b = c view, however, is not complete. The authors purposively chose a centralized model to deliver the intervention and acknowledged that this approach could be a potential contributor to the study’s insufficient effect. This approach did not allow for the investigation of the specific health system design attributes that may have contributed to the findings as well as community-based factors, the other essential components of the CCM (15,16). There is a further need to investigate such health system factors as absence of decision support, visit planning, and outreach systems as well as practice-based planning, coordination and the use of analyzed, actionable PGHD into the workflow (17).
In a year-long RCT of behavioral mobile coaching for metabolic, lifestyle, and self-management for people with T2D combined with analyzed PGHD integrated with evidence-based guidelines for the care team, there was an overall reduction in A1C of 1.9% compared to 0.7% in the control group (18). Also, a difference of 0.3% was noticed between the group that had the coaching only intervention compared with the group that shared the analyzed PGHD with their HCP, indicating that perhaps treatment intensification contributed to this difference (18). A post-hoc analysis demonstrated significant differences in HCPs making changes to diabetes medications in the intervention arm as compared to the control group (19).
The ability of digital technology to support PWD and other chronic conditions with real-time, contextual coaching as well as generate actionable, intelligent reports for HCPs to encourage medication titration and support behavior changes can decrease therapeutic inertia. These solutions provide the ability to capture lifestyle, behavioral, and metabolic data at the individual level that can then be summarized to a population level to improve HCP workflow. One caution, the volume of data generated with these solutions needs to be analyzed against evidence-based guidelines and presented to HCPs with suggested actions, so the data facilitates decision-making instead of introducing a new burden for the already challenged health care team. In 2019, new Current Procedural Terminology (CPT) codes for care coordination and remote monitoring will expand payment to the support staff and may help to incorporate these solutions into practice as well as begin to move the needle on outcomes (20).
Tackling the therapeutic inertia problem requires multiple approaches. The Endocrine Society is addressing one of the fears associated with medication management in T2D, the costly problem of hypoglycemia, by leading an initiative with the primary care community. This multi-year quality improvement project targets improved surveillance, best practices for insulin management, and engages patients in self-management (21). The American Heart Association has established its Precision Medicine Platform (22) to enable global research on cardiovascular and brain disease. Opportunities for learning more about the interaction of genetics, lifestyle, and environment will provide further insights into addressing therapeutic challenges.
There are 2 underlying factors that need to be addressed at the start of any new initiative or evaluation, understanding the social determinants of health (23) and the burden of treatment of the condition (24). HCPs and health systems need to recognize that PWD have different capacity for engaging in their health (23). All solutions need to be designed to decrease the burden for both patients and the care team (24).
Therapeutic inertia is a major challenge. It will take multi-faceted approaches to move the needle addressing both the patient and HCPs (6). This paper by Barton et al. has contributed further understanding to this complex, challenging problem. Organizational initiatives such as the ADA multi-year initiative, the Endocrine Society Hypoglycemia Prevention and the American Heart Precision Medicine Platform are opportunities for learning more about how to address this challenging problem. However, a simple focus on changing the language used when talking with and about PWD can go a long way to build trust needed to improve therapeutic inertia, and is simple and cost effective to begin immediately (7).
Acknowledgements
None.
Footnote
Conflicts of Interest: DA Greenwood consults for technology companies and Peeples is employed by a technology company. M Peeples has no conflicts of interest to declare.
References
- American Diabetes Association. Overcoming Therapeutic Inertia. 2018. Available online: https://professional.diabetes.org/meeting/other/overcoming-therapeutic-inertia
- Khunti K, Gomes MB, Pocock S, et al. Therapeutic inertia in the treatment of hyperglycaemia in patients with type 2 diabetes: A systematic review. Diabetes Obes Metab 2018;20:427-37. [Crossref] [PubMed]
- Davies MJ, D'Alessio DA, Fradkin J, et al. Management of Hyperglycemia in Type 2 Diabetes, 2018. A Consensus Report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 2018;41:2669-701. [Crossref] [PubMed]
- Barton AB, Okorodudu DE, Bosworth HB, et al. Clinical Inertia in a Randomized Trial of Telemedicine-Based Chronic Disease Management: Lessons Learned. Telemed J E Health 2018;24:742-8. [Crossref] [PubMed]
- Powers BJ, King JL, Ali R, et al. The Cholesterol, Hypertension, and Glucose Education (CHANGE) study for African Americans with diabetes: study design and methodology. Am Heart J 2009;158:342-8. [Crossref] [PubMed]
- Capoccia K, Odegard PS, Letassy N. Medication Adherence With Diabetes Medication: A Systematic Review of the Literature. Diabetes Educ 2016;42:34-71. [Crossref] [PubMed]
- Dickinson JK, Guzman SJ, Maryniuk MD, et al. The Use of Language in Diabetes Care and Education. Diabetes Care 2017;40:1790-9. [Crossref] [PubMed]
- Brown MT, Bussell J, Dutta S, et al. Medication Adherence: Truth and Consequences. Am J Med Sci 2016;351:387-99. [Crossref] [PubMed]
- Polonsky WH, Arsenault J, Fisher L, et al. Initiating insulin: How to help people with type 2 diabetes start and continue insulin successfully. Int J Clin Pract 2017.71. [PubMed]
- Digital Therapeutics Alliance. Available online: https://www.dtxalliance.org/dtx-solutions. Accessed December 10, 2018./
- Greenwood DA, Gee PM, Fatkin KJ, et al. A Systematic Review of Reviews Evaluating Technology-Enabled Diabetes Self-Management Education and Support. J Diabetes Sci Technol 2017;11:1015-27. [Crossref] [PubMed]
- Greenwood DA, Blozis SA, Young HM, et al. Overcoming Clinical Inertia: A Randomized Clinical Trial of a Telehealth Remote Monitoring Intervention Using Paired Glucose Testing in Adults With Type 2 Diabetes. J Med Internet Res 2015;17:e178. [Crossref] [PubMed]
- Greenwood DA, Young HM, Quinn CC. Telehealth Remote Monitoring Systematic Review: Structured Self-monitoring of Blood Glucose and Impact on A1C. J Diabetes Sci Technol 2014;8:378-89. [Crossref] [PubMed]
- Wagner EH. Chronic Disease Management: What will it take to improve care for chronic illness? Eff Clin Pract 1998;1:2-4. [PubMed]
- Allen JD, Curtiss FR, Fairman KA. Nonadherence, clinical inertia, or therapeutic inertia? J Manag Care Pharm 2009;15:690-5. [Crossref] [PubMed]
- Ross SA. Breaking down patient and physician barriers to optimize glycemic control in type 2 diabetes. Am J Med 2013;126:S38-48. [Crossref] [PubMed]
- Reach G, Pechtner V, Gentilella R, et al. Clinical inertia and its impact on treatment intensification in people with type 2 diabetes mellitus. Diabetes Metab 2017;43:501-11. [Crossref] [PubMed]
- Quinn CC, Shardell MD, Terrin ML, et al. Cluster-randomized trial of a mobile phone personalized behavioral intervention for blood glucose control. Diabetes Care 2011;34:1934-42. [Crossref] [PubMed]
- Quinn CC, Sareh PL, Shardell ML, et al. Mobile Diabetes Intervention for Glycemic Control: Impact on Physician Prescribing. J Diabetes Sci Technol 2014;8:362-70. [Crossref] [PubMed]
- American Medical Association. 2019 CPT codes offer new paths to payment for digital medicine. Available online: https://www.ama-assn.org/practice-management/cpt/2019-cpt-codes-offer-new-paths-payment-digital-medicine. Accessed December 14, 2018.
- Endocrine Society. Hypoglycemia Prevention Initiative. Available online: https://www.endocrine.org/hypoglycemia-prevention-initiative. Accessed December 12, 2018.
- American Heart Association. The Institute for Precision Cardiovascular Medicine. Available online: https://precision.heart.org. Accessed December 12, 2018.
- Clark ML, Utz SW. Social determinants of type 2 diabetes and health in the United States. World J Diabetes 2014;5:296-304. [Crossref] [PubMed]
- Tran VT, Barnes C, Montori VM, et al. Taxonomy of the burden of treatment: a multi-country web-based qualitative study of patients with chronic conditions. BMC Med 2015;13:115. [Crossref] [PubMed]
Cite this article as: Greenwood DA, Peeples M. Technology to overcome therapeutic inertia. mHealth 2019;5:1.